|
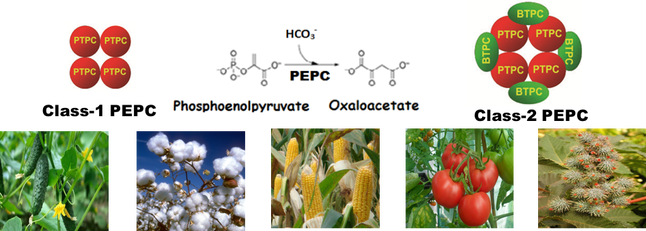
Transcriptome-Enabled Biochemistry: Unraveling the function of bacterial-type phosphoenolpyruvate carboxylase in vascular plants PEP carboxylase (PEPC) is a tightly regulated enzyme that plays essential roles in all plant cells, particularly the ‘anaplerotic’ replenishment of TCA cycle intermediates withdrawn for biosynthesis. Plant PEPC belongs to a small multigene family encoding several closely related plant-type PEPCs (PTPC) along with a distantly-related and enigmatic bacterial-type PEPC (BTPC). Previous characterization of PEPC from green microalgae and then developing castor beans led to the discovery of novel, Class-2 PEPC hetero-octameric complexes composed of tightly associated PTPC and BTPC subunits and that are largely desensitized to classic PEPC allosteric inhibitors such as malate. Mike’s bioinformatics analysis of publicly available RNA-seq and microarray transcriptomic datasets revealed two distinct patterns of tissue-specific BTPC expression. Species such as Arabidopsis, rice, maize, and poplar mainly exhibited pollen-specific BTPC expression. By contrast, BTPC transcripts were particularly abundant in non-pollen, heterotrophic ‘sink’ tissues including developing castor, cotton, soybean, and cucumber seeds, as well as the developing fruits of avocado, grape, and tomato. Follow up biochemical and co-immunoprecipitation experiments confirmed presence of ~118 kDa BTPC polypeptides that associate with co-expressed PTPC subunits to form a Class-2 PEPC complex in tomato and cucumber fruits. Overall results support the hypothesis that BTPC functions as a catalytic and regulatory subunit of Class-2 PEPCs that sustain rapid anaplerotic carboxylation of PEP while refixing respired CO2 in biosynthetically active sink tissues that accumulate abundant amounts of malate. Comments are closed.
|
Archives
February 2021
|